
You need to prepare everyone in the organization for the audit. If you need to achieve the ISO standard, you will go for an internal compliance audit and hence determine the assurance processes and management system that needs to be examined. 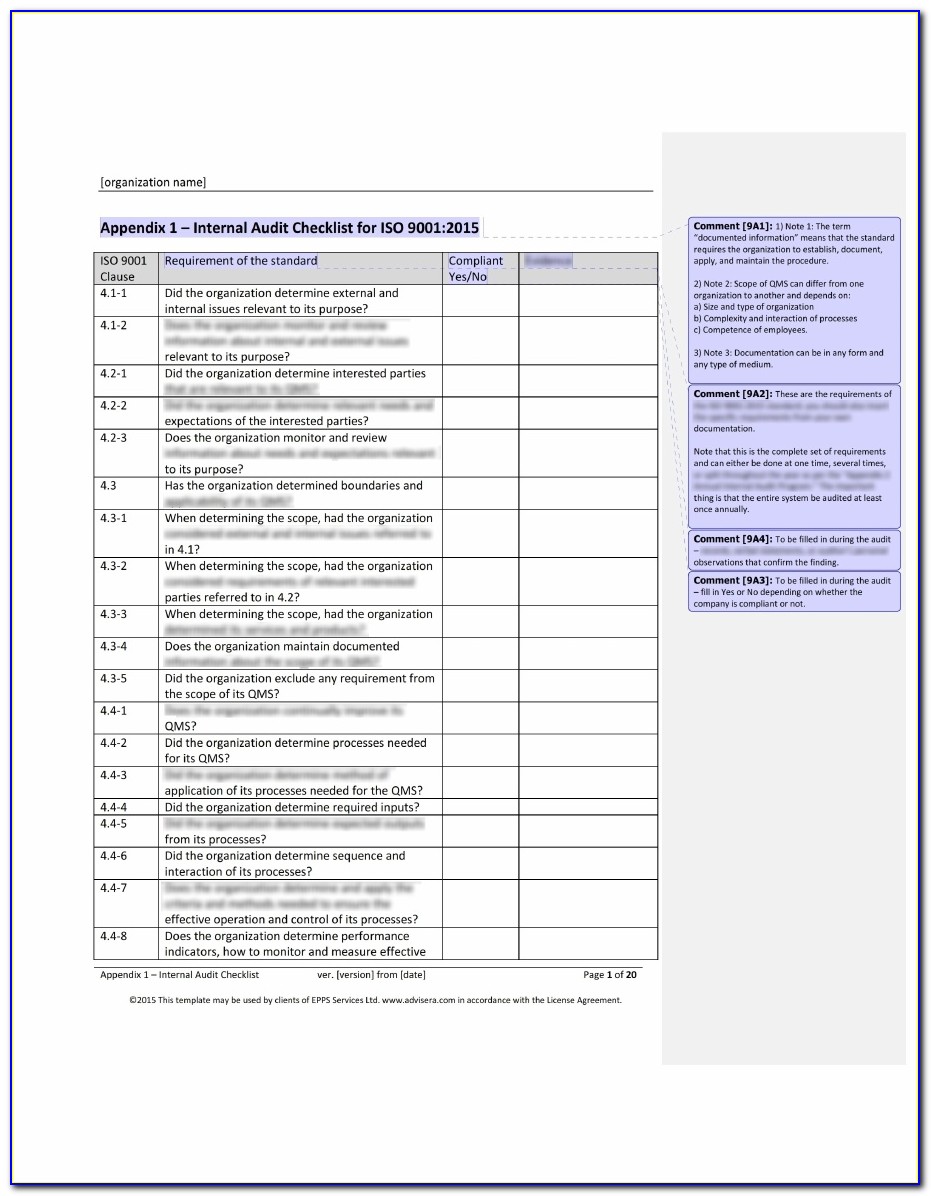
This clearly depends on your current goals or targets. Therefore, the first step is to decide the specific audit strategy needed in your medical organization at present. Then, there is a certification compliance audit which is conducted to check the conformity of quality management processes to the standard. It can be a product audit where only final products or services are evaluated or it can be a supplier audit where all the materials, inputs, or services used in the production are checked by an external third-party organization. An audit can be a process audit where the internal processes or systems of the organization are checked. Since there are different types and purposes for audits within an organization, you need to first decide what type of audit to conduct in your medical device company. Internal Audit Checklist for Medical Devices that Simplifies the Audit ProcedureĪudits may seem to be a time-consuming and overwhelming task but planning them properly and dividing the task into simple steps can reduce the complexity of the audit and the time required. Here we present the internal audit checklist which includes the simplified steps to carry out in the audit procedure. If your business operates in the medical device industry and is awaiting this ISO certification, this blog is for you. Audits are also an important way to improve the effectiveness of their quality management approach and further enhance the value of medical devices. While the audits can be challenging, following the internal audit checklist for medical devices will help to smoothly sail through the audit process. However, compliance requires rigorous internal audits of the quality management processes. Conforming compliance with the standard helps a medical device company to exhibit its stringent efforts for assuring the quality of medical devices and so enables them to secure a competitive place in the market. It is a specific standard that provides guidelines and requirements for the quality management framework in medical devices. Hundreds of manufacturers and suppliers in the medical device industry are getting the international quality management standard ISO 13485. CAPA also calls for measures to help mitigate and prevent future occurrences of the same quality issues.Step-by-Step Internal Audit Checklist for Medical Device Companies It’s not enough to correct nonconformances and other quality issues. Evaluate steps taken to resolve nonconformances.CAPA implementation should be formally documented and its records maintained. Implement correction of nonconformances and their causesĬorrective action and preventive action (CAPA) is a crucial aspect of ISO audit procedures.The documentation aspect of an internal audit program should include policies, procedures, instructions, and records of audits that have been performed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
